Dissolved inorganic carbon

Dissolved inorganic carbon (DIC) is the sum of the aqueous species of inorganic carbon in a solution. Carbon compounds can be distinguished as either organic or inorganic, and as dissolved or particulate, depending on their composition. Organic carbon forms the backbone of key component of organic compounds such as – proteins, lipids, carbohydrates, and nucleic acids.
Inorganic carbon is found primarily in simple compounds such as carbon dioxide, carbonic acid, bicarbonate, and carbonate (CO2, H2CO3, HCO−
3, CO2−
3 respectively). Dissolved inorganic carbon (DIC) includes three major aqueous species, CO2, HCO−
3 ,CO2−
3, and to a lesser extent their complexes in solution with metal ions.[3]
Marine ecosystems
[edit]| Part of a series on the |
| Carbon cycle |
|---|
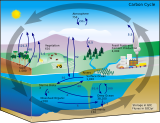 |
Solubility pump
[edit]Aqueous carbon dioxide reacts with water to form carbonic acid which is very unstable and will dissociate rapidly into hydronium and bicarbonate. Therefore, in seawater, dissolved inorganic carbon is commonly referred to as the collection of bicarbonate, carbonate ions, and dissolved carbon dioxide (CO2, H2CO3, HCO−
3, CO2−
3).
- CO2(aq) + H2O ⇌ H2CO3 ⇌ HCO−
3 + H+ ⇌ CO2−
3 + 2 H+
More than 99% of dissolved inorganic carbon is in the form of bicarbonate and carbonate ions meaning that most of the ocean’s carbon storing ability is due to this chemical reactivity.[4] Sea-air flux of CO2 and the resulting dissolved inorganic carbon is affected by physical processes such as strong winds and vertical mixing, and the biological processes of photosynthesis, respiration, and decomposition.[5]
Biological pump
[edit]Dissolved inorganic carbon is a key component of the biological pump, which is defined as the amount of biologically produced organic carbon flux from the upper ocean to the deep ocean.[6] Dissolved inorganic carbon in the form of carbon dioxide is fixed into organic carbon is produced through photosynthesis. Respiration is the reverse process and consumes organic carbon to produce inorganic carbon. Photosynthesis, and the biological pump, is dependent on the availability of inorganic nutrients and carbon dioxide.[7]
- Photosynthesis: 6 CO2 + 6 H2O + light → C6H12O6 + 6 O2
- Respiration: C6H12O6 + 6 O2 → 6 CO2 + 6 H2O + energy
Oceanographers seek to understand the metabolic state of the ocean, or the efficiency of the biological pump, by estimating the net community production (NCP) which is the gross primary productivity (GPP) minus the community respiration (sum of the respiration of the local autotrophs and heterotrophs).[8] An efficient biological pump increases biological export to the deeper ocean which has been hypothesized to suppress CO2 outgassing in the upper ocean.[9][10]

Carbonate pump
[edit]The carbonate pump is sometimes referred to as the “hard tissue” component of the biological pump.[12] Some surface marine organisms, like Coccolithophores, produce hard structures out of calcium carbonate, a form of particulate inorganic carbon, by fixing bicarbonate.[13] This fixation of DIC is an important part of the oceanic carbon cycle.
- Ca2+ + 2 HCO−
3 → CaCO3 + CO2 + H2O
While the biological carbon pump fixes inorganic carbon (CO2) into particulate organic carbon in the form of sugar (C6H12O6), the carbonate pump fixes inorganic bicarbonate and causes a net release of CO2.[14] In this way, the carbonate pump could be termed the carbonate counter pump. It works counter to the biological pump by counteracting the CO2 flux from the biological pump.
Measurement
[edit]Oceanographers and engineers continue to find novel and more accurate methods of measuring carbon content in seawater. One method is to collect water samples and directly measure the DIC by using a TOC analyzer.[15] Samples can be combined with stable isotope ratios 13C/12C, alkalinity measurements, and estimation of physical processes, to create diagnostic techniques.[16] Researchers at Scripps Institution of Oceanography developed a tool that uses flow injection analysis to measures microfluidic samples of seawater and continuously monitor dissolved inorganic carbon content.[17]
See also
[edit]- Alkalinity (total alkalinity; AT)
- Bjerrum plot
- Dissolved organic carbon
- Fugacity (carbon dioxide fugacity; fCO2)
- Ocean acidification
- pH
- Total organic carbon
References
[edit]- ^ Environmental Research at Tanguro Ranch, Brazil Esri. Retrieved 26 July 2020.
- ^ Neu, V., Ward, N.D., Krusche, A.V. and Nill, C. (2016) "Dissolved organic and inorganic carbon flow paths in an Amazonian transitional forest". Frontiers in Marine Science, 3: 114. doi:10.3389/fmars.2016.00114.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
- ^ Mackenzie FT and Lerman A (2006) Carbon in the Geobiosphere: Earth's Outer Shell, Springer Science & Business Media. ISBN 9781402042386.
- ^ Williams, Richard G.; Follows, Michael J. (14 July 2011). Ocean Dynamics and the Carbon Cycle: Principles and Mechanisms. Cambridge, UK: Cambridge University Press. pp. 7–9. ISBN 978-0521843690.
- ^ Ma, W; Chai, F; Xiu, P; Tian, J (2014). "Simulation of export production and biological pump structure in the South China Sea". Geo-Marine Letters. 34 (6): 541–554. Bibcode:2014GML....34..541M. doi:10.1007/s00367-014-0384-0. S2CID 129982048.
- ^ Emerson, Steven (2014). "Annual net community production and the biological carbon flux in the ocean". Global Biogeochemical Cycles. 28 (1): 14–28. Bibcode:2014GBioC..28...14E. doi:10.1002/2013GB004680.
- ^ Raymond, Peter A.; Bauer, James E. (2000). "Atmospheric CO2 evasion, dissolved inorganic carbon production, and net heterotrophy in the York River estuary". Limnol. Oceanogr. 45 (8): 1707–1717. Bibcode:2000LimOc..45.1707R. doi:10.4319/lo.2000.45.8.1707.
- ^ Ducklow, H.W.; Doney, S.C. (2013). "What is the Metabolic State of the Oligotrophic Ocean? A Debate". Annual Review of Marine Science. 5: 525–33. doi:10.1146/annurev-marine-121211-172331. hdl:1912/5282. PMID 22809191.
- ^ Ma, W; Chai, F; Xiu, P; Tian, J (2014). "Simulation of export production and biological pump structure in the South China Sea". Geo-Marine Letters. 34 (6): 541–554. Bibcode:2014GML....34..541M. doi:10.1007/s00367-014-0384-0. S2CID 129982048.
- ^ Kim, H.J.; Kim, T.-W; Hyeong, K; Yeh, S.-W.; Park, J.-Y.; Yoo, C.M.; Hwang, J. (2019). "Suppressed CO2 Outgassing by an Enhanced Biological Pump in the Eastern Tropical Pacific". Journal of Geophysical Research: Oceans. 124 (11): 7962–7973. Bibcode:2019JGRC..124.7962K. doi:10.1029/2019JC015287. S2CID 210611691.
- ^ Wu, Y., Hain, M.P., Humphreys, M.P., Hartman, S. and Tyrrell, T. (2019) "What drives the latitudinal gradient in open-ocean surface dissolved inorganic carbon concentration?" Biogeosciences, 16(13): 2661–2681. doi:10.5194/bg-16-2661-2019.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
- ^ Hain, M.P.; Sigman, D.M.; Haug, G.H (2014). "The Biological Pump in the Past". Treatise on Geochemistry. 8: 485–517. doi:10.1016/B978-0-08-095975-7.00618-5. ISBN 9780080983004.
- ^ Rost, Bjorn; Reibessel, Ulf (2004). Coccolithophores and the biological pump: responses to environmental changes. Berlin, Heidelberg: Springer. ISBN 978-3-642-06016-8.
- ^ Rost, Bjorn; Reibessel, Ulf (2004). Coccolithophores and the biological pump: responses to environmental changes. Berlin, Heidelberg: Springer. ISBN 978-3-642-06016-8.
- ^ Raymond, Peter A.; Bauer, James E. (2000). "Atmospheric CO2 evasion, dissolved inorganic carbon production, and net heterotrophy in the York River estuary". Limnol. Oceanogr. 45 (8): 1707–1717. Bibcode:2000LimOc..45.1707R. doi:10.4319/lo.2000.45.8.1707.
- ^ Gruber, Nicolas; Kneeling, Charles D.; Stocker, Thomas F. (1998). "Carbon-13 constraints on the seasonal inorganics carbon budget at the BATS site in the northwestern Sargasso Sea". Deep-Sea Research Part I. 45 (4–5): 673–717. Bibcode:1998DSRI...45..673G. doi:10.1016/S0967-0637(97)00098-8.
- ^ Bresnahan, Philip J.; Martz, Todd R. (2018). "Gas Diffusion Cell Geometry for a Microfluidic Dissolved Inorganic Carbon Analyzer". IEEE Sensors Journal. 8 (6): 2211–2217. Bibcode:2018ISenJ..18.2211B. doi:10.1109/JSEN.2018.2794882. S2CID 3475999.


